Florida is offering free monoclonal antibody treatment for COVID. What to know about it
More than a dozen state-run centers have opened across Florida offering free monoclonal antibody treatment to people sick with COVID-19 and considered to be high risk for severe illness or death.
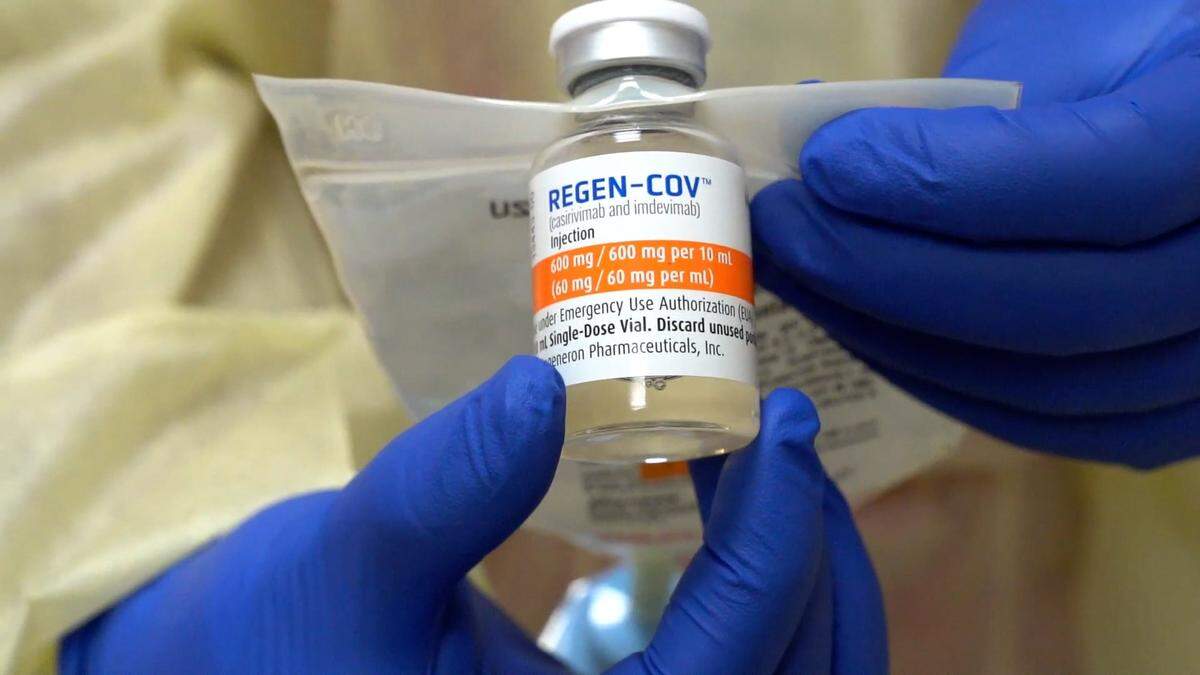
Often, people who visit these centers feel really sick. The therapy given to them — REGEN-COV by the biotechnology company Regeneron — is designed to give the immune system an extra boost to help combat the disease, enough to hopefully keep them out of the hospital.
The treatment, like Moderna’s and Johnson & Johnson’s COVID-19 vaccines, is under emergency-use authorization.
However, not everyone is eligible for the antibody cocktail.
Here’s what to know:
How do monoclonal antibody treatments work?
Monoclonal antibodies are laboratory-made “substitute antibodies” that can help the immune system recognize and respond more effectively to COVID-19, according to the U.S. Food and Drug Administration.
Florida’s state-run centers are using Regeneron’s antibody cocktail, which can be given through infusion or injection. It’s the therapy that former President Donald Trump received when he fell ill with COVID-19.
The monoclonal antibody treatment has shown to be effective against the more contagious delta variant that is now the predominant strain circulating in the United States, including in South Florida, as the Miami Herald has reported.
Regeneron’s antibody cocktail is meant to treat mild or moderate COVID-19 within 10 days of a person’s first symptom. Preliminary research shows it helped reduce the risk of hospitalization and death by 70%, according to the Washington Post.
The sooner that high-risk people get the treatment after testing positive, the better, said Dr. Kenneth Scheppke, the state EMS medical director for the Florida Department of Health and the chief medical officer for the Florida Division of Emergency Management.
Who can get Regeneron’s antibody cocktail in Florida and who should not?
The treatment is for people 12 and older, regardless of vaccination status, who have tested positive and are considered to be “high risk” for severe COVID illness, including hospitalization and death. High-risk people 12 and older who were exposed to COVID and are either not fully vaccinated or have an immunocompromised condition that has lowered their immune response to the vaccine are also eligible.
Some of the conditions the FDA considers to be high risk include diabetes, obesity, chronic lung disease (including moderate to severe asthma), and cardiovascular disease (including congenital heart disease) or hypertension.
Pregnancy, having an immunosuppressive disease or undergoing an immunosuppressive treatment, and being older than 65 are also considered high-risk conditions. The Centers for Disease Control and Prevention has a list of additional conditions that could make someone high risk for severe COVID infection.
Some people should not get Regeneron’s antibody cocktail, including people who are hospitalized for COVID-19 or require oxygen therapy due to the disease, according to the FDA.
Speak with your doctor about any safety or eligibility concerns you may have before getting the treatment.
Are monoclonal antibody treatments an alternative to COVID-19 vaccines? Can I get vaccinated afterward?
No, the treatment is not a substitute for vaccination. And yes, people can still get vaccinated after receiving a monoclonal antibody treatment, but they have to wait at least 90 days to do so, Scheppke said.
COVID-19 vaccines are still the most effective way in reducing the risk of falling ill with COVID-19 and preventing severe illness or death.
Think of it this way: Monoclonal antibodies help people currently ill with COVID-19. Vaccines train our immune system to fight off a future infection. COVID-19 vaccines also provide longer protection, according to the University of Alabama at Birmingham.
Pfizer’s vaccine was given the FDA’s full authorization in August for people 16 and older. The vaccine is still under emergency-use authorization for kids 12 to 15. The Moderna and Johnson & Johnson vaccines, which are for people 18 and older, are still under emergency-use authorization.
What are REGEN-COV’s side effects?
Possible side affects include nausea, diarrhea, dizziness, headache, itchiness and vomiting, according to the FDA’s REGEN-COV FAQ guide. Rare severe allergic reactions have also been reported in clinical studies.
“REGEN-COV is still being studied, so it is possible that all of the risks are not known at this time,” the FDA says.
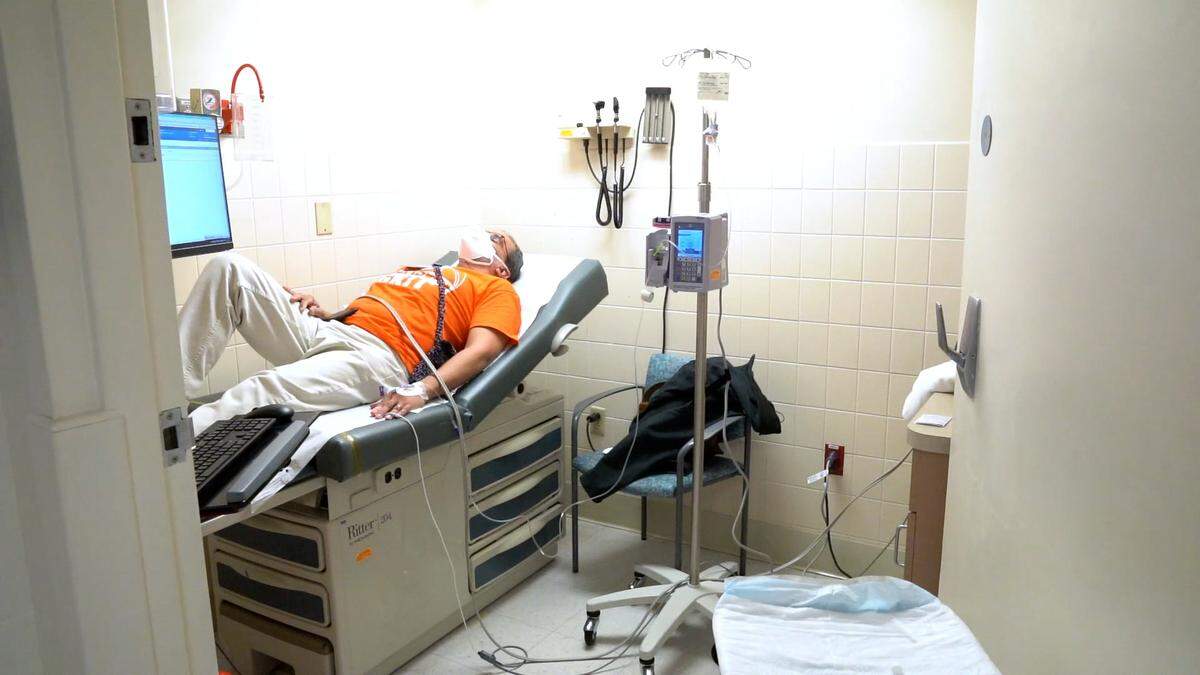
I’m visiting a monoclonal antibody center. What should I expect?
REGEN-COV consists of two investigational medicines, casirivimab and imdevimab, given together at the same time through an IV infusion or four injections. Patients will have to wait at the site for about an hour to make sure they don’t get side effects.
People who visit the center should expect to be there for several hours. All of the sites are climate-controlled and have seats and cots for people to lie down, Scheppke said.
Where can I get a monoclonal antibody treatment in Florida?
All of the state-run centers in Florida are open daily from 9 a.m. to 5 p.m. Appointments can be scheduled online. Treatment is free and can be given to eligible patients, regardless of vaccination status. No prescription or doctor referral is needed, according to the Florida Department of Health.
Some Florida hospitals may also be offering monoclonal antibody treatments. Urgent-care centers and private doctors are also authorized to provide the treatment, according to the Washington Post.
