Here’s why 11,000 bottles of ear infection medication have been recalled nationwide
Taro Pharmaceuticals recalled two lots of prescription medicine used for ear infections Wednesday after testing found a problem with an impurity and another with cortisone volume, according to an FDA Enforcement Report.
Here are the details on the recall of 11,196 bottles:
What ear medicine is recalled and why is it usually prescribed?
Taro yanked lot Nos. AC86809 and AC86812 of Hydrocortisone 1% and Acetic Acid 2% Otic Solution USP, National Drug Code No. 51672-3007-1. Both lots have expiration date 01/31/2024. Distributed was nationwide.
According to the National Library of Medicine, this is used for “treatment of superficial infections of the external auditory canal.” Stanford Medicine Children’s Health says that’s “the tube that connects the outer ear to the inside or middle ear.”
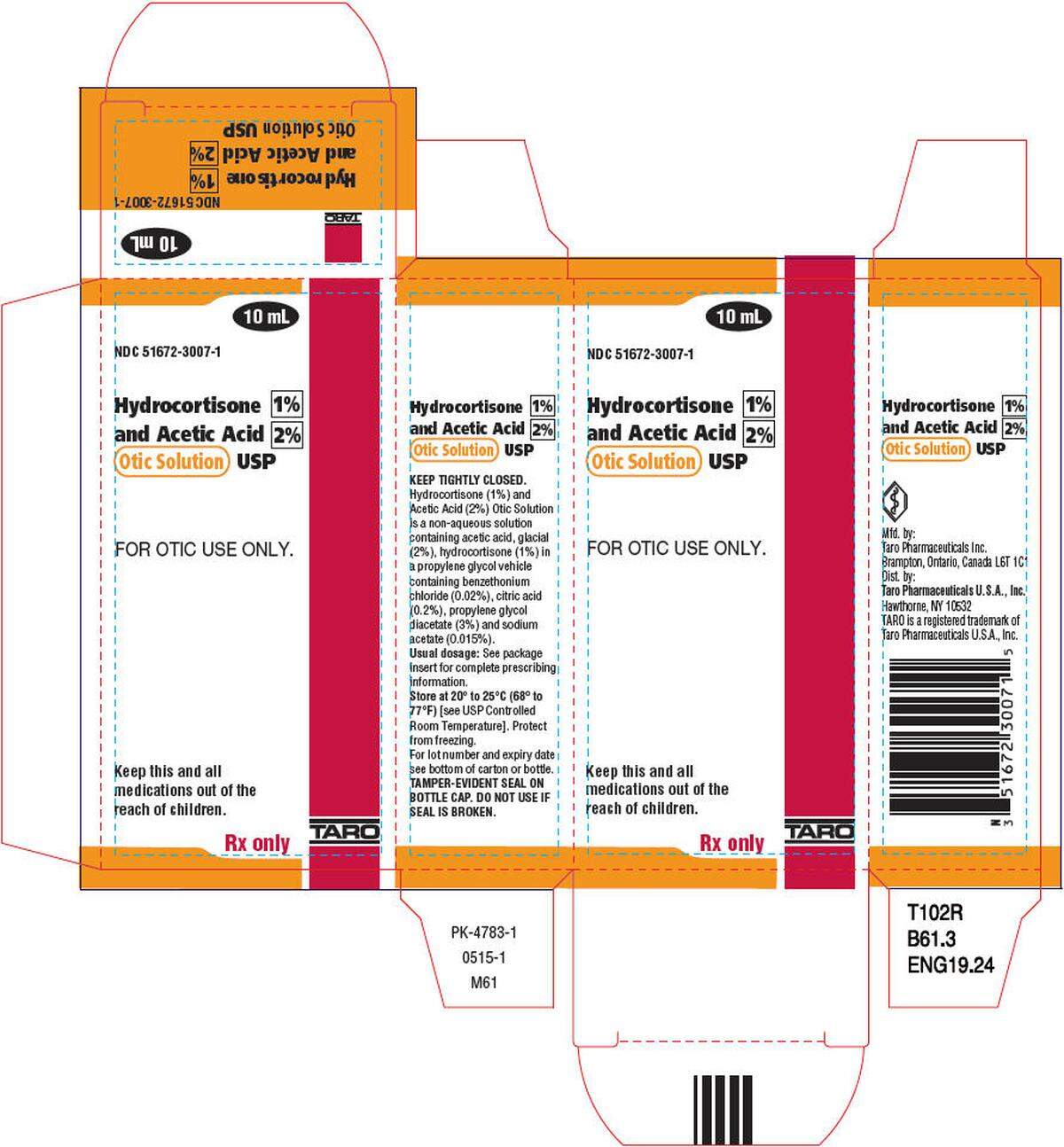
Why did Taro recall the Hydrocortisone and Acetic Acid?
The enforcement report explains, “Failed Impurities/Degradation Specifications: Out-of-Specification result for Hydrocortisone-related impurity and slightly lower than the established level of the Hydrocortisone assay obtained during stability testing.”
Translation: Testing revealed too much of an impurity and too little of the pain and swelling relief part of the drug. Both were out of the acceptable ranges.
“A confirmed [out-of-specification] result indicates that the batch does not meet established standards or specifications and should result in the batch’s rejection,” the FDA said in its guidance for dealing with out-of-specification test results.
What should you do now?
If you have a box from either lot, return it to the pharmacy or medical professional, then ask for a box from a lot that hasn’t been recalled or for a refund. Questions about this recall should be directed to Taro’s U.S. headquarters at 914-345-9001 or Canadian headquarters (the medicine was made in Brampton, Ontario) at 905-791-8276 (TARO).
Report any medical problems, first, to a medical professional. Then tell the FDA’s MedWatch program, either online or by calling 888-463-6332 (INFO-FDA) and pressing “2” when prompted.
This story was originally published November 7, 2023 at 11:43 AM.