Recall: This COVID-19 home test was illegally imported and not authorized by the FDA
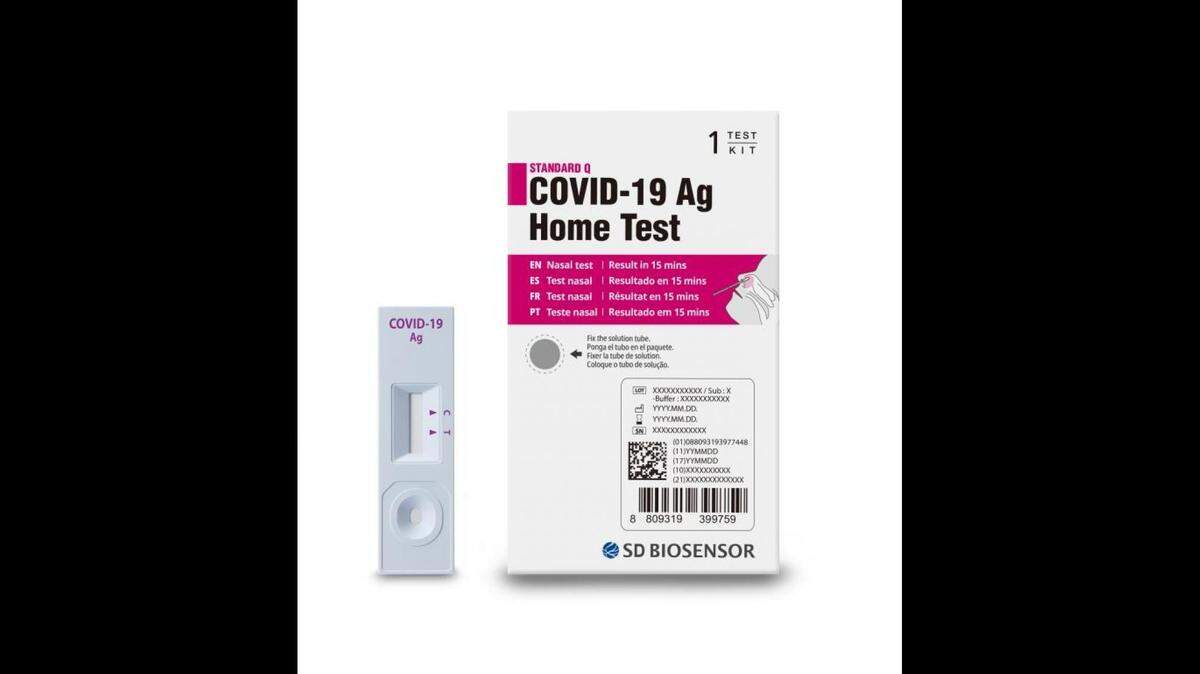
South Korean diagnostics company SD Biosensor recalled its Standard Q COVID-19 Ag Home Test from the United States after “confirmed reports that the test kits were illegally imported into the United States.”
That’s in the company-written, FDA-posted recall notice that says because the test hasn’t been “authorized, cleared or cleared or approved by the FDA” for U.S. use, consumers who have used it are “strongly encouraged to consider retesting with an FDA authorized or cleared test.”
SD Biosensor says “there is no known distribution of these tests directly to consumers” but it also says its only started the investigation into how the product got illegally imported to the United States.
“SD Biosensor, Inc. is taking appropriate measures to prevent further attempts at illegal importation of unauthorized tests by strengthening contract terms and their enforcement with its distributors,” the recall notice says. “In addition, the company announced publicly that if such illegal importations are discovered in the future, the responsible individuals/distributors will face strict legal action and liabilities for damages.”
This story was originally published February 5, 2022 at 9:02 AM.
