Mom runs out of formula, so flight attendant breast feeds passenger’s baby on flight
Flight attendants get called on to do all sorts of things in the air.
From dealing with unruly passengers, to bringing food and drink, and sometimes even to landing the plane as the case in 2016 when a flight attendant on a Boeing 747 with 195 people on board helped land the plane when the co-pilot passed out somewhere over France.
Now, meet the flight attendant who breast fed a stranger’s baby inflight when the passenger couldn’t.
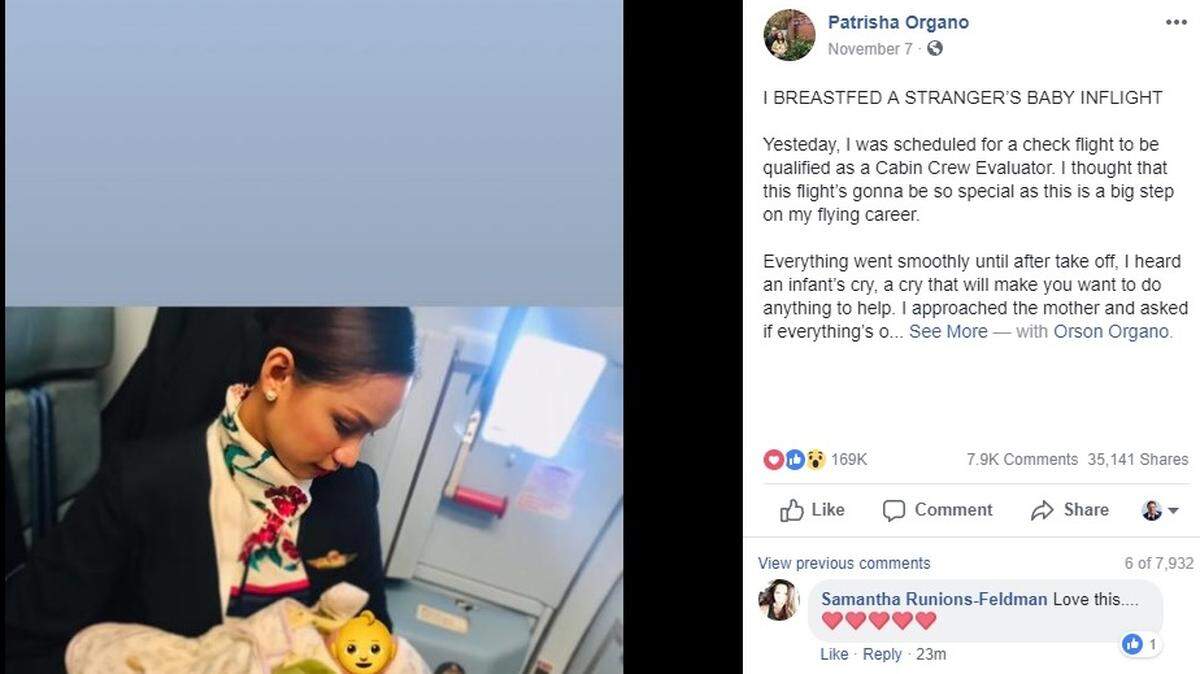
“Everything went smoothly until after take off,” Phillippine Airlines attendant Patrisha Organo wrote on her Facebook page on Wednesday about a domestic flight.
“I heard an infant’s cry, a cry that will make you want to do anything to help,” Organo wrote. “I approached the mother and asked if everything’s OK, I tried to tell her to feed her hungry child. Teary-eyed, she told me that she ran out of formula milk. Passengers started looking and staring at the tiny, fragile crying infant. I felt a pinch in my heart. There’s no formula milk on board. I thought to myself, there’s only one thing I could offer and that’s my own milk. And so I offered.”
Organo, 24, is a mom to a 9-month-old baby, the New York Post reported . Her Facebook post featured a photo of her holding the passenger’s baby, but she edited the picture to obscure the baby’s face so as to protect “her family’s privacy.”
The post was shared more than 35,000 times by Saturday afternoon.
“Love, love, love this,” read one of the comments. “Heartwarming,” read another.
But not everyone was on board with the idea of breast feeding another mother’s baby.
“The gesture was beautiful of the flight attendant ... but you do not know her health issues,” read a comment on Organo’s Facebook thread.
Indeed, the U.S. Food and Drug Administration warns that giving a baby breast milk from another mother can pose health risks.
“Risks for the baby include exposure to infectious diseases, including HIV, to chemical contaminants, such as some illegal drugs, and to a limited number of prescription drugs that might be in the human milk, if the donor has not been adequately screened,” the FDA counsels.
