One lot of a drug has been recalled for a cross contamination with lidocaine
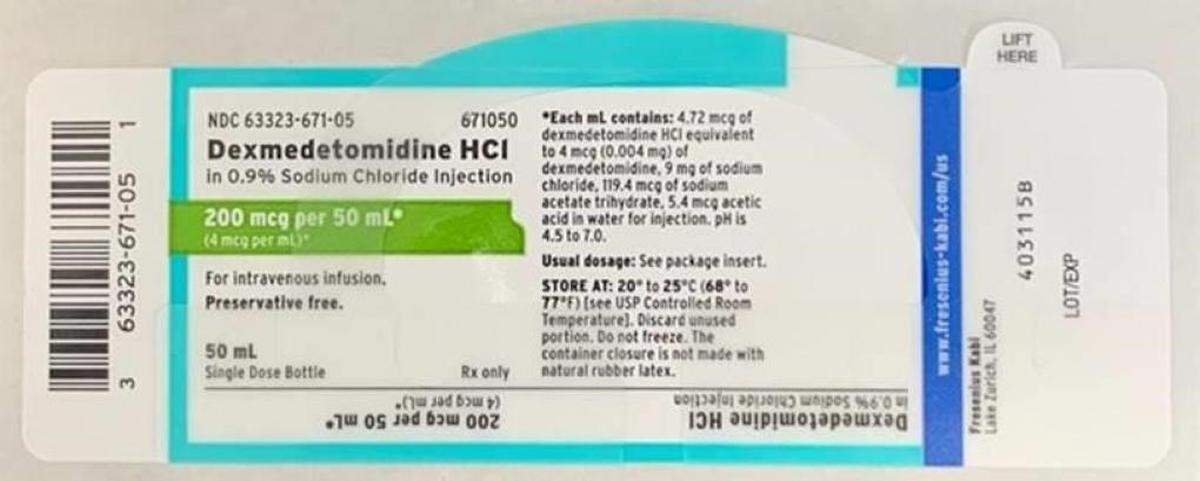
Trace amounts of lidocaine caused Fresenius Kabi USA to recall a batch of Dexmedetomidine HCl in 0.9% Sodium Chloride Injection.
The Fresenius-written, FDA-posted recall notice says, “Administration of Dexmedetomidine HCl containing trace amounts of lidocaine to a patient with lidocaine allergy could result in a potentially life-threatening allergic reaction.”
Dexmedetomidine HCl is a drug given intravenously before or during medical procedures, including surgeries.
What’s recalled went to wholesalers, distributors, hospitals and pharmacies April 9-13 and has batch No. 6123925 with an expiration date of March 2022. For questions about this recall, call Fresenius at 866-716-2459, Monday through Friday, 9 a.m. to 6 p.m., Eastern time.
If you suffer a medical problem from this or any other drug, you should first contact a medical professional. Then report the problem to the FDA’s MedWatch Adverse Event Reporting program either via the FDA website or by a form obtained at 800-332-1088.