The medicine didn’t dissolve as it should. That can be a serious problem
Eight lots of Tetracycline capsules that might not dissolve properly have been recalled by Avet Pharmaceuticals.
In the company-written, FDA-posted recall notice, Avet said the capsules’ didn’t perform properly in dissolution tests. Low dissolution means less medicine available to do the job for which it’s being taken.
“For patients with compromised immune systems and the elderly, who may be taking tetracycline to treat a serious infection such as pneumonia,” the notice says, “there is a reasonable probability that if there is not enough tetracycline in the body to fight the infection, this could result in rapid progression of the infection and death.”
Avet distributed the capsules in 100-count bottles under the Heritage Pharmaceuticals brand name.
The recalled 250 mg bottles had NDC No. 23155-017-01, lot no. H190666 with an expiration date of July 2022.

The seven lots of 500 mg capsules had NDC No. 23155-018-01, lot Nos. G190609, G190610, G190611 with expiration date of June 2022; lot Nos. L191027 and L191028 with an expiration date of November 2022; and lot Nos. K190953 and K190952 with an expiration date of October 2022.
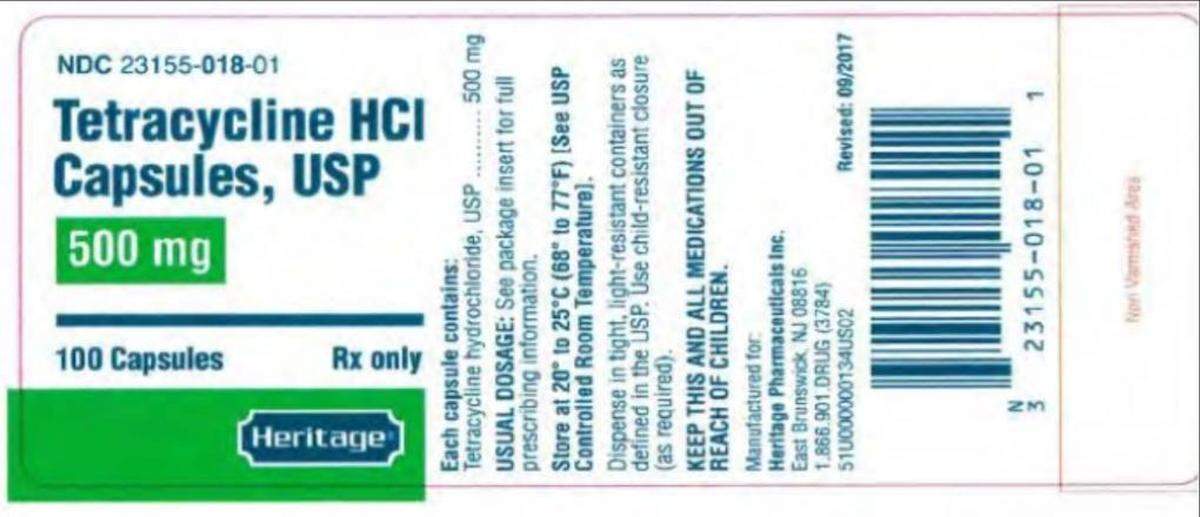
Avet advises that patients taking these capsules talk to a doctor or healthcare professional before stopping. Those with questions can contact Qualanex, which is handling the recall for Avet, at 888-424-4341, Monday through Friday, 8 a.m. to 5 p.m., Eastern time.
