The type of pills on the bottle’s label weren’t the type of pills in the bottle
No one wants a surprise in their pill bottles. So when Accord Healthcare heard about a 100-count bottle of hydrochlorothiazide 12.5 mg tablets instead contained 100 spironolactone Tablets USP 25 mg, the4 company recalled the entire lot.
“Since the individual lot, PW05264, of the product is involved in a potential mix-up of labeling, Accord is recalling this individual lot from the market,” said the company-written, FDA-posted recall notice. “Based on findings of both preliminary and interim investigations carried out at the manufacturing site, Accord believes that no other lots of hydrochlorothiazide Tablets are involved in this mix-up.”
A customer complained to the pharmacy, which then informed Accord.
Spironolactone can be used to treat heart problems, high blood pressure, low potassium levels and fluid retention for some maladies, according to emedicinehealth.com. Hydrochlorothiazide treats high blood pressure and fluid retention, says rxlist.com.
“Use of spironolactone tablets instead of hydrochlorothiazide tablets, poses the risk of contracting hyperkalemia (increased potassium levels) in certain individuals resulting in adverse events that range from limited health consequences to life-threatening situations in certain individuals,” the notice explains.
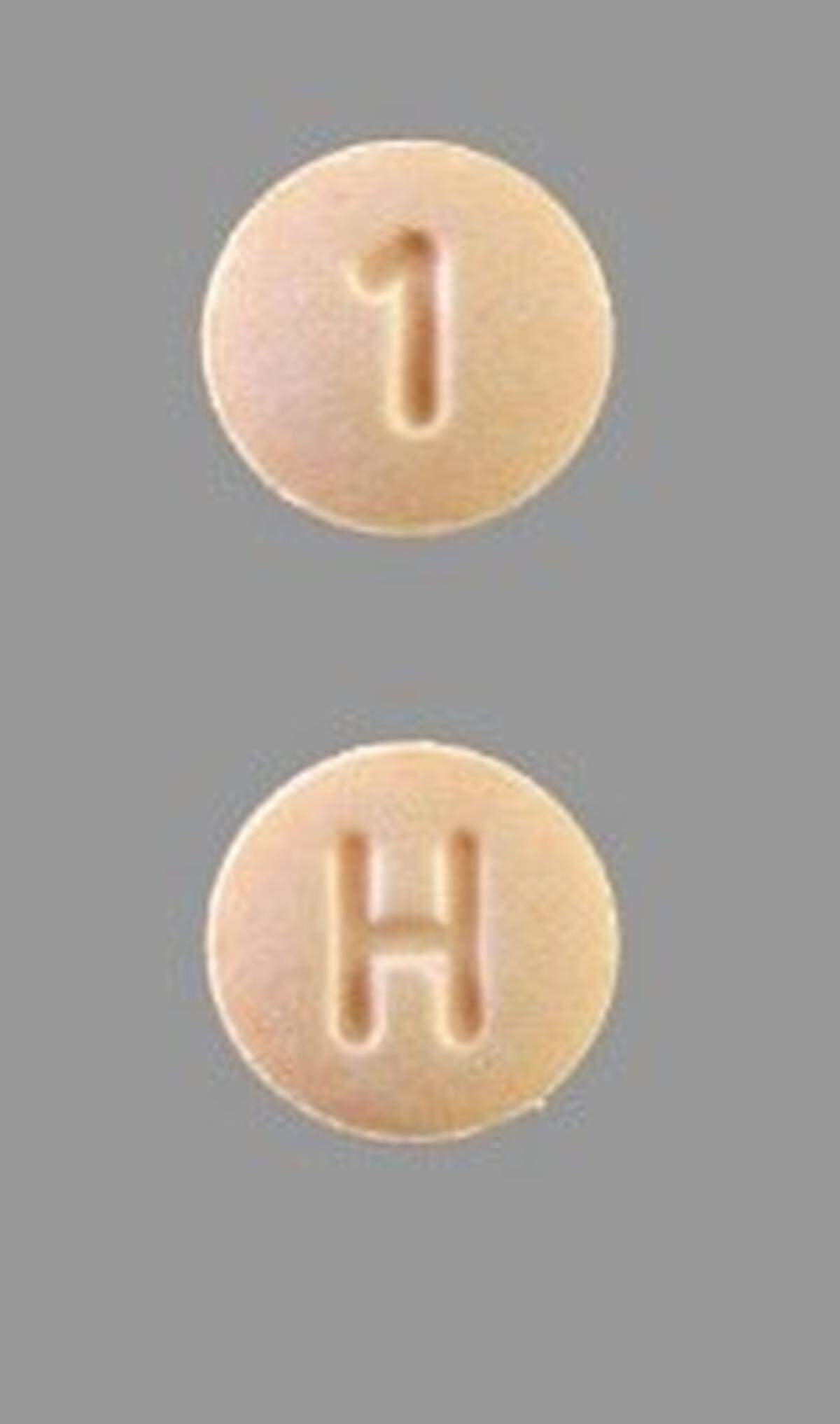
Hydrochlorothiazide 12.5 mg pills are orange circles with an “H” on one side and a “1” on the other. Anyone unsure if the pills in their hydrochlorothiazide bottle are hydrochlorothiazide should take them back to their pharmacist or to a healthcare professional.
Anyone with questions can contact Accord at 855-869-1081, by fax at 1-817-868-5362, or by emailing rxrecalls@inmar.com, Monday through Friday, from 8 a.m. to 5 p.m, Eastern time.
This story was originally published August 29, 2018 at 6:25 AM.