Drugstore’s sinus spray recalled over a ‘microbiological contamination’
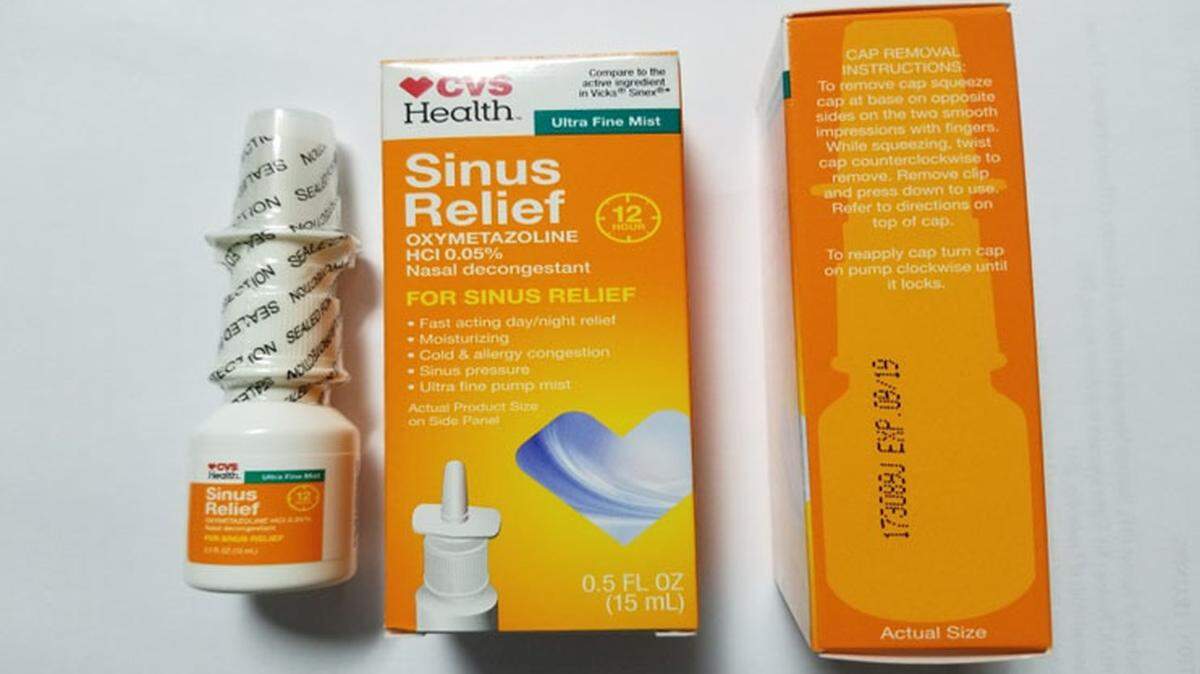
One lot of CVS 12-Hour Sinus Relief Nasal Mist has been recalled because of a “microbiological contamination identified as Pseudomonas aeruginosa,” manufacturer Product Quest announced Wednesday in an FDA-posted recall notice.
The notice from the Holly Hill, Florida-based company reads, “Repetitive use of a nasal spray containing a gram-negative pathogen can potentially lead to colonization and subsequent infection which can be life threatening in certain patient populations, such as those with cystic fibrosis or (with damaged immune systems).”
Lot No. 173089J with an expiration date of 09/19 was distributed to CVS stores nationwide. Consumers with nasal spray from the recalled lot should return it for a refund or toss it out.
Those with questions can call Product Quest at 386-239-8787, Monday through Friday, 8 a.m. to 4 p.m., Eastern time.
“Serious Pseudomonas infections usually occur in people in the hospital and/or with weakened immune systems. Infections of the blood, pneumonia, and infections following surgery can lead to severe illness and death in these people,” the Centers for Disease Control explains. “However, healthy people can also develop mild illnesses with Pseudomonas aeruginosa, especially after exposure to water.
“Ear infections, especially in children, and more generalized skin rashes may occur after exposure to inadequately chlorinated hot tubs or swimming pools. Eye infections have occasionally been reported in persons using extended-wear contact lenses.”
This story was originally published August 8, 2018 at 4:06 PM.