Crushed by demand, monoclonal antibody treatment sites close in Miami-Dade and Broward
With the omicron variant of the coronavirus spreading rapidly across South Florida and the number of new COVID-19 cases in the region surging again, state-run monoclonal antibody treatment sites in Miami-Dade and Broward were forced to temporarily close on Tuesday after running out of supplies.
CDR Health, the healthcare company hired by the state to provide testing, vaccination and monoclonal antibody treatments in Florida, postponed an undetermined number of appointments in Miami-Dade and Broward, and at sites in Lee and St. Lucie counties, on Tuesday because “we have nothing there,” said Steve Vancore, a spokesman.
“We’ve had to temporarily close those sites simply because we have no supplies, which means all the people who had appointments are being called,” Vancore said.
But the Florida Department of Health said the sites were closed because they were “training staff in additional administration techniques,” Weesam Khoury, communications director, said in a prepared statement. She said the four sites would reopen on Wednesday, and that the agency was also “reallocating monoclonals from areas of the state with less demand to expand services.”
Khoury said the therapies were still available Tuesday through alternative providers, including Broward Health North, the public hospital system for North Broward; Lee Memorial Hospital in Fort Myers; and Longwood Regional Medical Center in Fort Pierce.
She did not provide an alternative site for Miami-Dade, where patients had been receiving monoclonal antibody treatments at Tropical Park in Miami since about August and which had long lines of people early Tuesday morning waiting for the site to open.
‘An urgent shortage’
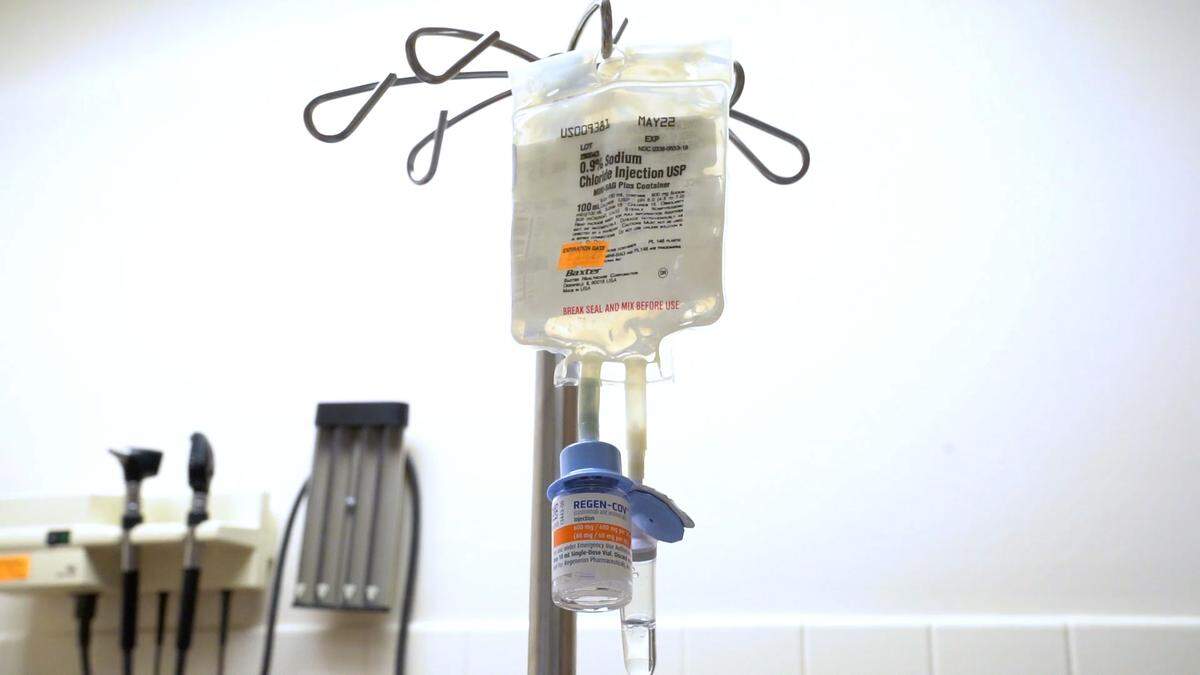
To date, Miami-Dade’s site has administered more than 18,400 doses of the monoclonal antibody treatment called REGEN-COV, which is effective against the delta variant but “completely lost inhibitory activity” against the highly mutated omicron strain, according to recent research conducted by the University of Washington School of Medicine and published online ahead of peer review.
CDR Health is expected to resume administering monoclonal antibody treatments in Tropical Park on Wednesday but the company will be using the therapy called Bamlanivimab, which research has shown to be ineffective against the delta variant that is still circulating in Florida.
Like REGEN-COV, Bamlanivimab also is reportedly ineffective against omicron, according to the University of Washington study.
On Tuesday, Miami-Dade Mayor Daniella Levine Cava urged the state’s Surgeon General, Dr. Joseph Ladapo, to help ensure that Miami-Dade receives monoclonal antibody therapies now that cases are soaring in the county and the rate of COVID-19 tests returning positive has reached 12% — indicating a high rate of transmission.
“Like other parts of the state, we are now facing an urgent shortage of monoclonal antibody treatment. ... Given Miami-Dade is the epicenter of the spread of Omicron with some of the highest positivity in the state, we urge you to direct all possible resources to our County to ensure we can provide this lifesaving treatment to our residents.”
Vancore, the CDR Health spokesman, said the company’s supply of monoclonal antibodies from the state was “very fluid.”
“It’s literally a day-by-day thing,” he said. “In some regards, it’s an hour-by-hour thing.”
Cases rising, omicron dominant
Miami-Dade residents can access monoclonal antibody treatments at some local providers, including Jackson Health System, the county’s public hospital network.
But a Jackson Health spokeswoman confirmed Tuesday that there’s a shortage of the REGEN-COV monoclonal antibody treatment, and that early evidence indicates that particular drug may be less effective against the omicron variant, which is now the predominant strain of the coronavirus circulating in Miami-Dade and the United States.
“Jackson Health System is regularly updating its clinical guidance about when to provide this form of therapy,” said Tania Leets, a spokeswoman.
Khoury did not respond to questions about whether Florida is experiencing a statewide shortage of monoclonal antibody treatments due to a surge in demand. But cases are rising rapidly in the state.
On Tuesday, the Centers for Disease Control and Prevention reported the largest single-day increase in COVID-19 cases for Florida since Sept. 21 — 12,915 people confirmed with the disease.
Monoclonal antibody treatments are one of the few effective therapies available for COVID-19. The treatments are distributed by the federal government through the Department of Health and Human Services, which has contracted with the manufacturers to buy all the treatments.
HHS officials did not respond to questions Tuesday about the supply of monoclonal antibodies available for states. But on Dec. 17, the agency issued a statement that the federal government would begin shipping one type of at least four FDA-authorized monoclonal antibody treatments to Florida: sotorivimab, the only one that appears to work for the omicron variant but which is in short supply.
The federal government said it has 55,002 doses of sotorivimab available for all states and has allocated 1,050 doses for Florida, which is reporting thousands of new COVID-19 cases a day. Additional shipments of sotorivimab, which is manufactured by GlaxoSmithKline and received the FDA’s emergency use authorization in May, will not be available until the week of Jan. 3.
Guidance on monoclonal antibodies
Khoury said Florida has requested additional allocations of monoclonal antibodies from the federal government, but she did not respond to questions about whether the state has received its allocation of sotorivimab or how the agency would prioritize which patients receive the drug first.
HHS has advised states to preserve sotorivimab for eligible patients infected with the omicron variant and to use other monoclonal antibody therapies, including Eli Lilly’s Bamlanivimab/etesevimab, Regeneron’s REGEN-COV and AstraZeneca’s EvuSheld, all of which have been available in Florida.
The federal government also advised providers to preserve sotorivimab for patients in communities where the reported prevalence of omicron is greater than 20%. But that guidance already appears to be out of date despite being issued on Dec. 17.
On Monday, Miami-Dade officials reported that the omicron variant had been detected in 76% of 504 COVID-19 test samples taken on Dec. 14-15 — a dramatic increase from 1.3% of 373 samples collected Dec. 1-5. The CDC also announced that the highly mutated strain first identified in in November in South Africa now accounts for 73% of all cases in the United States.
Monoclonal antibodies are laboratory-made molecules that act as substitute antibodies. Delivered by injection or infusion, they can help a patient’s immune system recognize and respond more effectively to the coronavirus, making it more difficult for the virus to reproduce and cause harm.
Monoclonal antibody therapies are not intended as a substitute for vaccination, and the treatments can be limited in their effectiveness.
Some, such as REGEN-COV, have not been shown to benefit infected persons once they are hospitalized, and another, EvuSheld, which received the FDA’s emergency use authorization on Dec. 8, is intended as a prevention for COVID-19 in certain individuals with weakened immune systems and allergy to vaccines.
Another limiting factor is only one monoclonal antibody therapy — sotorivimab — has been found effective against the omicron variant. In a study published online on Dec. 17 before peer review, researchers found that several but not all monoclonal antibodies would fail to neutralize the omicron variant.
‘Waiting for data’
Dr. Aldo Calvo, who oversees monoclonal antibody therapy treatments for Broward Health, said physicians do not yet know whether the current batch of treatments made by Regeneron, Eli Lilly and others will work against the new variant. The hospital has not received any doses of sotorivimab.
“We are still waiting for data on the effectiveness of the current monoclonal antibodies against the Omicron variant,” Calvo said in a prepared statement. “When it comes to the Delta variant, the current treatment is 70% effective in reducing hospitalizations and deaths when given within 72 hours of the onset of symptoms. Currently, sotorivimab has not been made available. We are hopeful that we’ll receive our share.”
Khoury, the health department spokeswoman, noted that as Florida’s health department continues to monitor disease severity from an omicron infection, “the symptoms are still relatively mild.”
However, even if the omicron variant causes a less severe illness than prior strains of the coronavirus, its extreme contagiousness and ability to avoid immunity from vaccines or prior infection means many more people will get infected, raising the likelihood that more vulnerable people with weakened immune systems, cancer and other high-risk conditions will be hospitalized.
By the time a patient is admitted to a hospital for COVID-19, it is too late for the therapy because monoclonal antibodies are not authorized for use in patients who are hospitalized, according to the most recent COVID-19 treatment guidelines from the National Institutes of Health.
Feds cap supply
The first monoclonal antibody treatments received the FDA’s emergency use authorization in November 2020, and for months the demand was lower than supply.
In August, Gov. Ron DeSantis launched a statewide effort to increase patient access to the therapies with four state-run sites in Jacksonville, Orlando, Merritt Island and Broward County. More sites followed in Miami-Dade and Palm Beach counties, and as of Tuesday Florida’s health department listed at least 26 locations in the state where patients can access monoclonal antibodies, though that list includes the four that were closed on Tuesday.
But Florida’s access to monoclonal antibodies was limited in September, when the Biden administration changed the allocation method for the drugs, requiring hospitals and other facilities to work through their state governments to obtain the medications. Previously, the federal government had allowed hospitals and other healthcare providers to order monoclonal antibodies directly from the supplier.
Once the federal government limited the state’s supply, Florida could no longer directly obtain the treatments as the state had done previously.
Khoury, the health department spokeswoman, said the state is dependent on the federal government for supply and has requested additional allocations in light of the omicron variant’s increased contagiousness.
This story was originally published December 21, 2021 at 8:13 PM.
