The dosage on two lots of this prescription pain medication can cause you to overdose
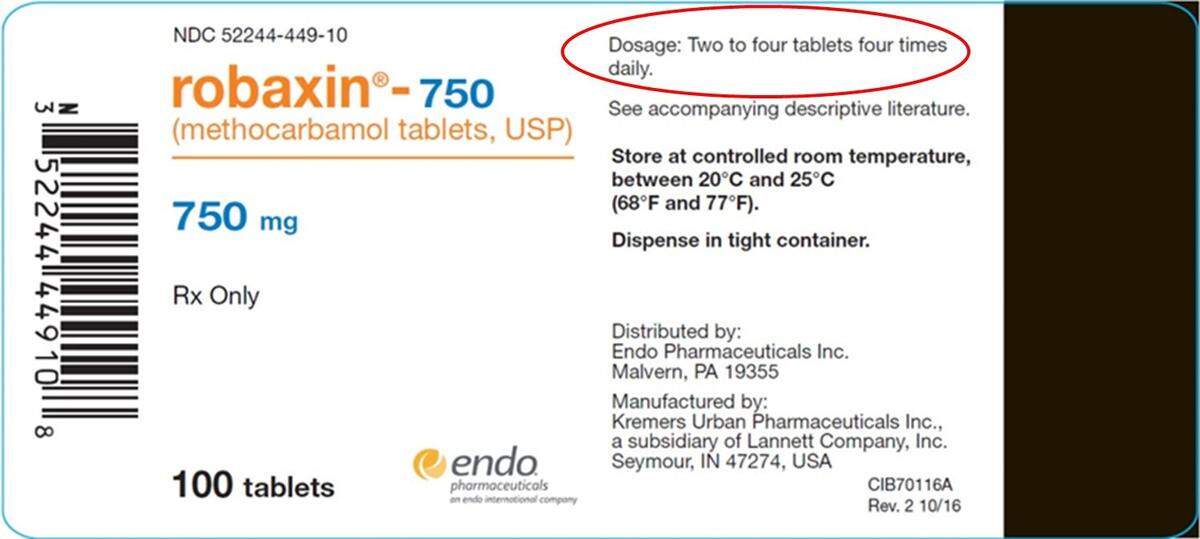
Endo Pharmaceuticals has recalled two lots of Robaxin pain medication nationwide because the label carries the wrong dosage, an amount that could cause an overdose.
The label in the recalled lots says take “two to four tablets four times daily.” The correct dosage, according to Endo’s recall announcement, is two tablets three times daily.
Lot Nos. 216702P1 with an expiration date of September 2020 and 220409P1 with an expiration date of January 2021 are both 100-count bottles of 750mg tablets.
Distributors and retailers should return any bottles from these lots to the wholesaler or distributor of sale. Any distributors or retailers with questions should e-mail robaxin@inmar.com or call 866-391-0620, Monday through Friday, 9 a.m. to 5 p.m., Eastern time.
Endo instructs consumers with the recalled Robaxin to stop using it and return it. Call 866-391-0620 or e-mail robaxin@inmar.com for instructions on returning product. For reimbursement, send proof of purchase to robaxin@inmar.com or via regular mail to Inmar, Attn. Recall Department, Robaxin Recall, 635 Vine St., Winston Salem, NC, 27101.
This story was originally published October 3, 2018 at 9:47 AM.